Industry News
Research, Science & Manufacturer Updates
FDA Updates Articles
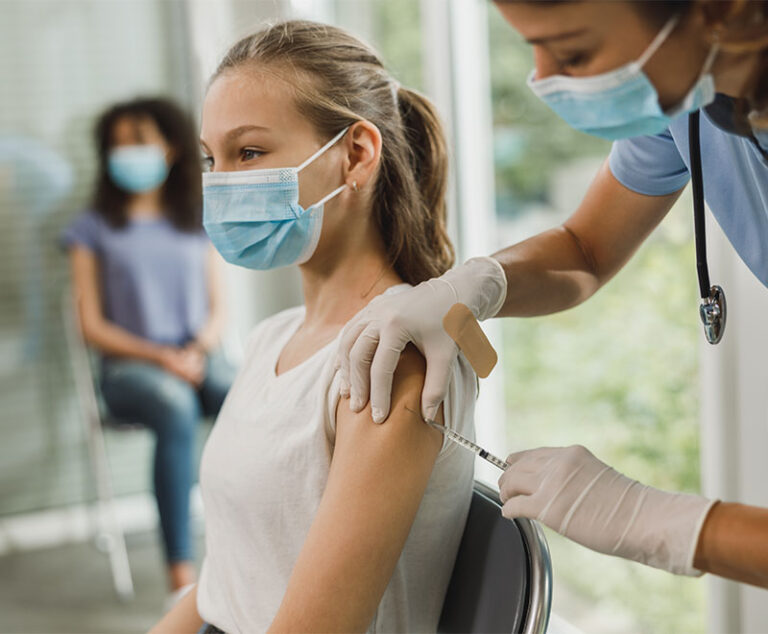
GSK’s Penmenvy (meningococcal groups A, B, C, W and Y vaccine) has been approved by the U.S. Food and Drug Administration for use in individuals aged 10 through 25 years.
The U.S. Food and Drug Administration (FDA) has expanded its approval of Dupixent to chronic obstructive pulmonary disease (COPD).
FDA has approved and granted emergency use authorization for updated mRNA COVID-19 vaccines (2024-2025 formula) to include a monovalent (single) component that corresponds to the Omicron variant KP.2 strain of SARSCoV-2.
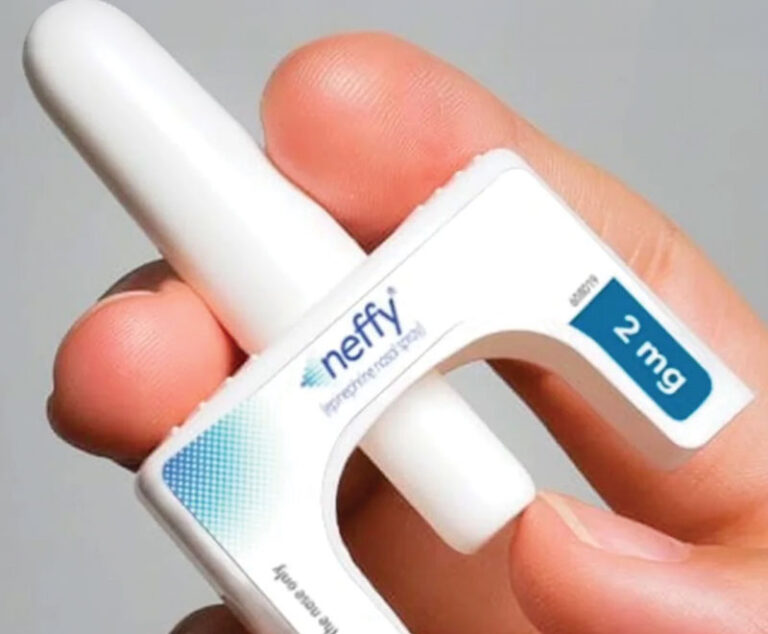
The U.S. Food and Drug Administration (FDA) has approved the first needle-free alternative to the EpiPen.
FDA has approved Octapharma USA's Fibryga (fibrinogen [human] lyophilized powder for reconstitution) for fibrinogen replacement in bleeding patients with acquired fibrinogen deficiency.
FDA has published new draft guidance on informed consent that lines up with revisions to the Common Rule made in 2017, offering up-to-date recommendations on starting the process with the sharing of essential clinical trial information in ways that patients can understand.
The U.S. Food and Drug Administration has amended the emergency use authorizations of the. Moderna and Pfizer-BioNTech COVID-19 bivalent mRNA vaccines to simplify the vaccination schedule for most individuals.
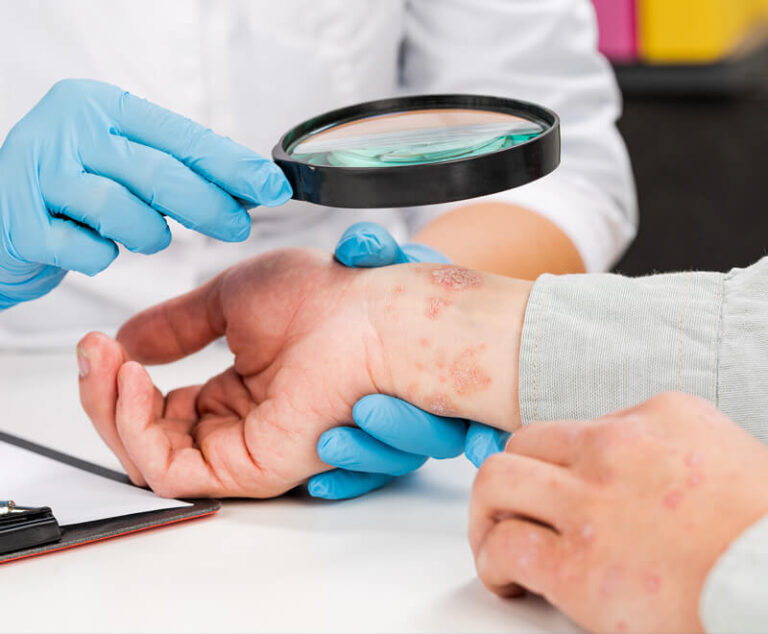
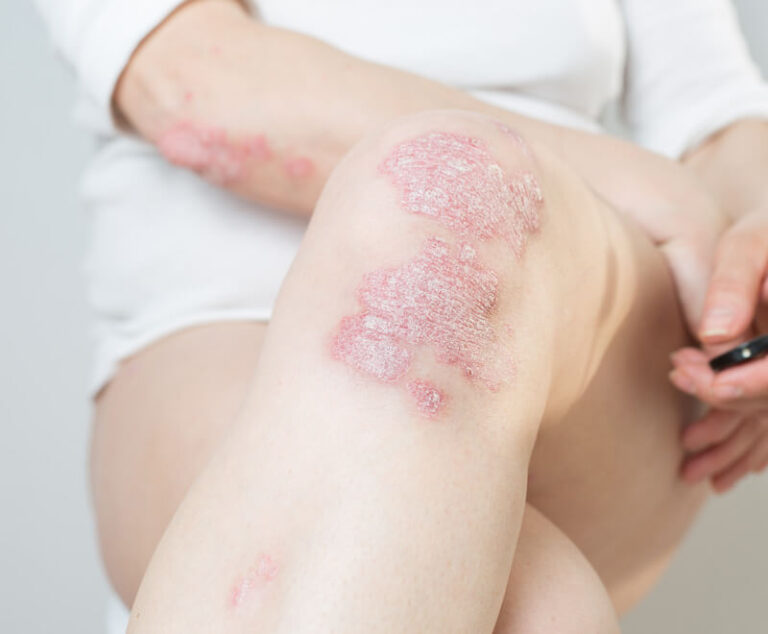
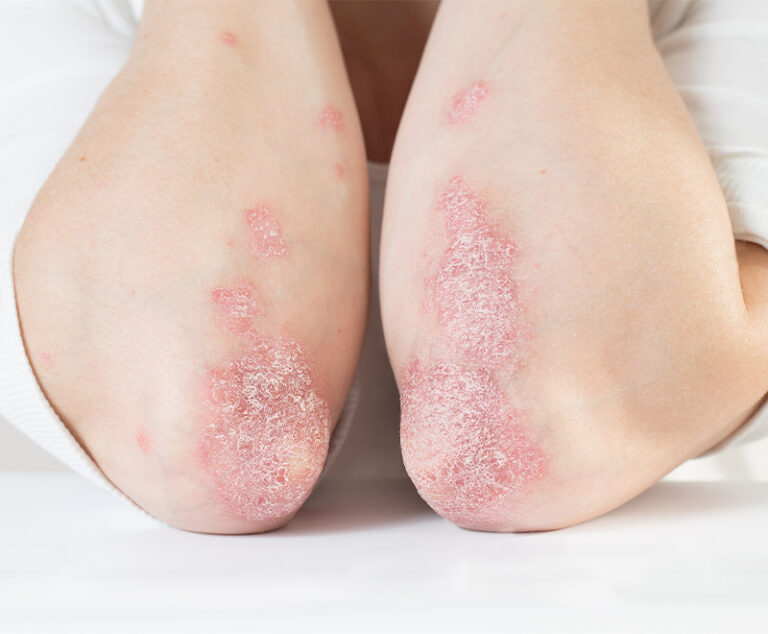
The U.S. Food and Drug Administration has approved Sotyktu (deucravacitinib), a once-daily oral pill by Bristol Myers Squibb, for adults who have plaque psoriasis (a chronic, systemic, immune-mediated disease) severe enough to make them candidates for systemic therapy and phototherapy.
The U.S. Food and Drug Administration has approved Samsung Bioepis; Halima (adalimumab-bwwd) for the treatment of several autoimmune disorders, including rheumatoid arthritis, psoriatic arthritis, ulcerative colitis and Crohn's disease.
AbbVie’s Rinvoq and Pfizer’s Cibinqo have been approved by the U.S. Food and Drug Administration (FDA) to treat moderate-to-severe atopic dermatitis.
The U.S. Food and Drug Administration (FDA) has approved Vyvgart (efgartigi-mod) to treat generalized myasthenia gravis (gMG) in adults who test positive for the anti-acetylcholine receptor (AChR) antibody.
The U.S. Food and Drug Administration (FDA) has approved Chemo- Centryx Inc.’s Avacopan, sold under the brand name Tavneos, to treat antineutrophil cytoplasmic antibody-associated vasculitides.