Industry News
Research, Science & Manufacturer Updates
Medicines Articles
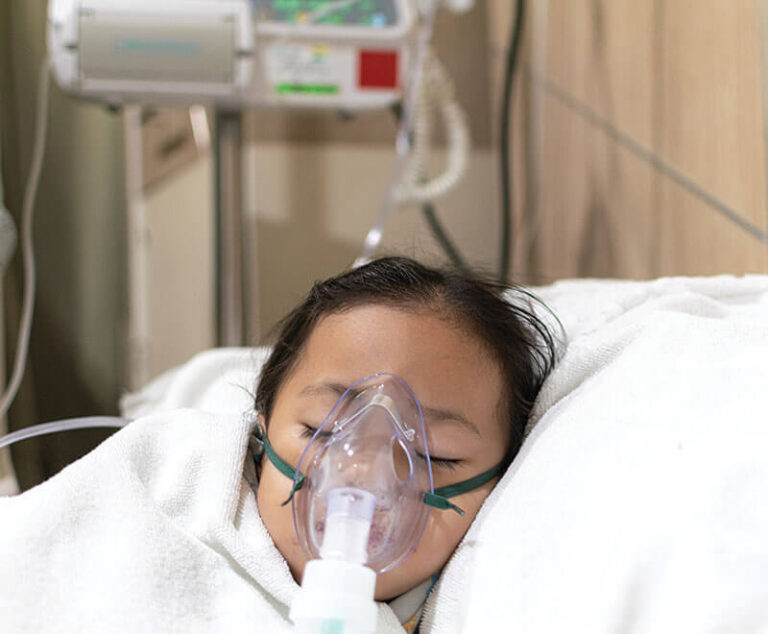
Pfizer's ABRYSVO, a respiratory syncytial virus (RSV) vaccine, has been approved by the U.S. Food and Drug Administration for the prevention of lower respiratory tract disease (LRTD) and severe LRTD caused by RSV in infants from birth up to 6 months of age by active immunization of pregnant women at 32 through 36 weeks gestational age.
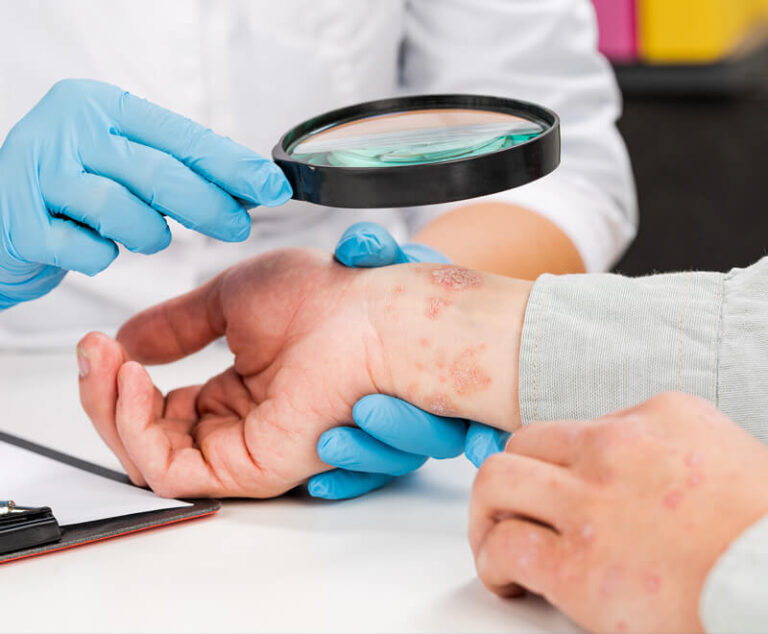
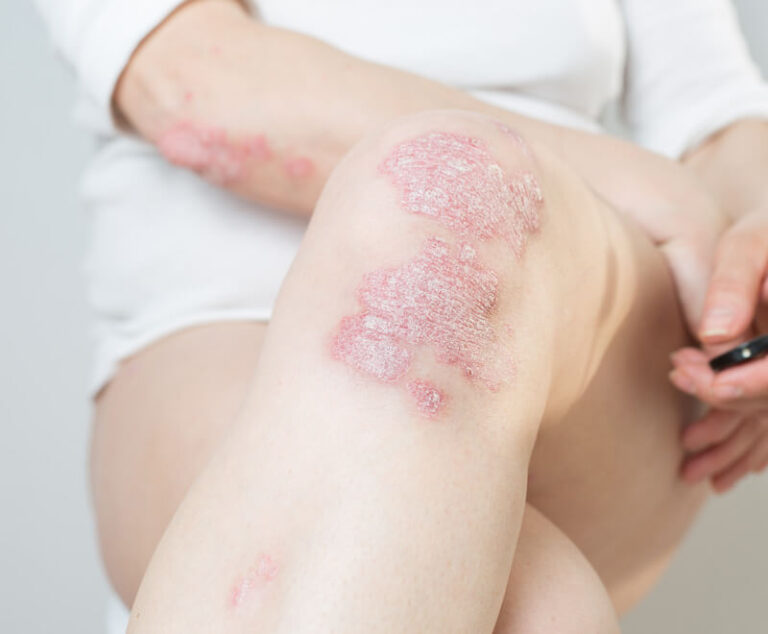
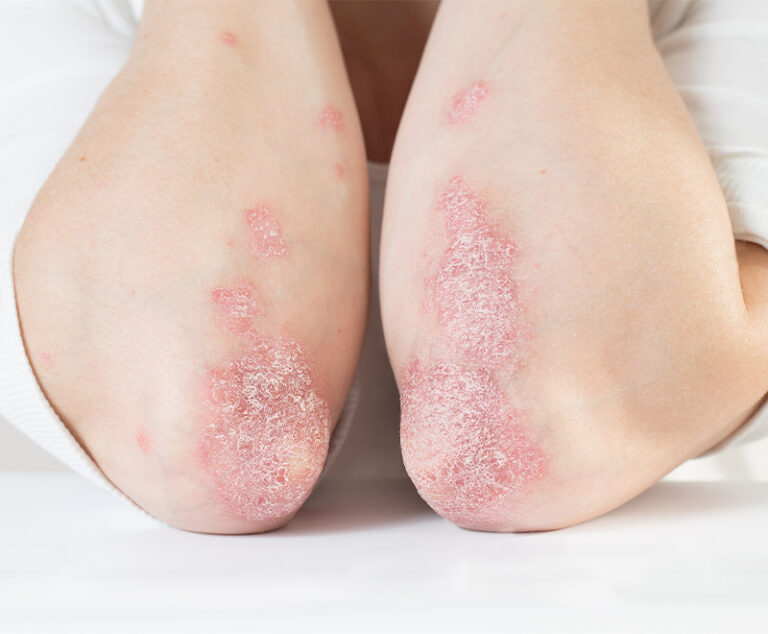
The U.S. Food and Drug Administration has approved Sotyktu (deucravacitinib), a once-daily oral pill by Bristol Myers Squibb, for adults who have plaque psoriasis (a chronic, systemic, immune-mediated disease) severe enough to make them candidates for systemic therapy and phototherapy.
The U.S. Food and Drug Administration has approved Samsung Bioepis; Halima (adalimumab-bwwd) for the treatment of several autoimmune disorders, including rheumatoid arthritis, psoriatic arthritis, ulcerative colitis and Crohn's disease.
Dutch investigators conducted a systematic review and meta-analysis of studies on the effectiveness of IVIG treatment of this specific population.
A new systematic review and meta-analysis conducted by U.S. and Nepalese collaborators supports the use of IVIG with glucocorticoids compared to IVIG alone.
AbbVie’s Rinvoq and Pfizer’s Cibinqo have been approved by the U.S. Food and Drug Administration (FDA) to treat moderate-to-severe atopic dermatitis.
Novartis has received U.S. Food and Drug Administration (FDA) approval for its Cosentyx (secukinumab) to treat children and adolescents with enthesitis-related arthritis and psoriatic arthritis.
While intravenous immune globulin (IVIG) therapy is efficacious for patients with chronic inflammatory demyelinating polyneuropathy (CIDP), the lack of biomarkers for disease activity makes the need for ongoing treatment difficult to assess.
Eltrombopay found to improve therapy for aplastic anemia.
Observational studies suggest immune globulin (IG) treatment may reduce the frequency of acute exacerbations of chronic obstructive pulmonary disease (AECOPD).
A large multicenter clinical trial has found intravenous immune globulin (IVIG) plus glucocorticoids may be better than IVIG alone for treating multisystem inflammatory syndrome in children (MIS-C) caused by COVID-19.
Kedrion Biopharma is now marketing and distributing RYPLAZIM (plasminogen human-tvmh) in the United States to treat C-PLGD.