Industry News
Research, Science & Manufacturer Updates
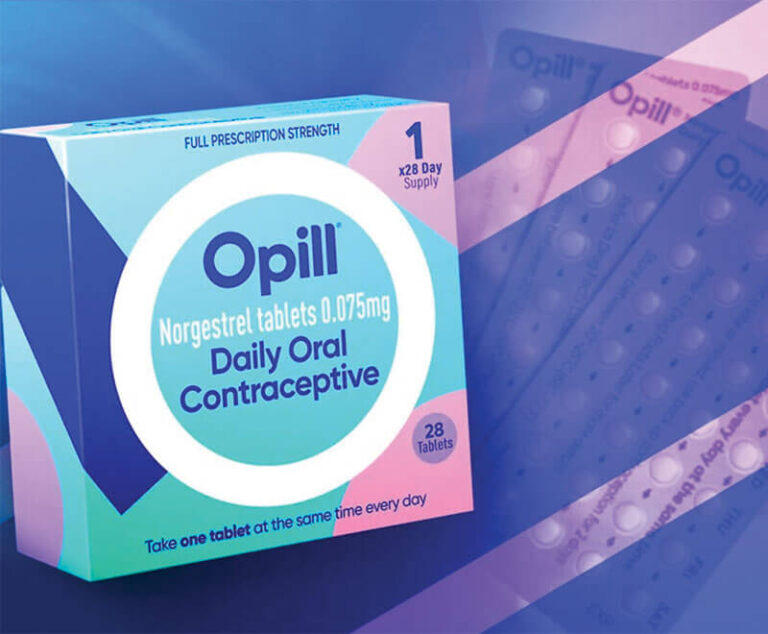
The U.S. Food and Drug Administration has approved the birth control pill Opill (norgestrel), manufactured by Perrigo, to be available over-the-counter — the first nonprescription birth control pill in the United States.
Pfizer's ABRYSVO, a respiratory syncytial virus (RSV) vaccine, has been approved by the U.S. Food and Drug Administration for the prevention of lower respiratory tract disease (LRTD) and severe LRTD caused by RSV in infants from birth up to 6 months of age by active immunization of pregnant women at 32 through 36 weeks gestational age.
The U.S. Food and Drug Administration (FDA) has granted 510(k) clearance to Precision BioLogic’s CRYOcheck Factor VIII (FVIII) Deficient Plasma with von Willebrand factor (VWF), opening the pathway for the company to launch the product in the U.S.
Researchers from the Pritzker School of Molecular Engineering at the University of Chicago have developed a new type of vaccine called an “inverse vaccine that completely reversed autoimmune diseases without fully shutting down the rest of the immune system.
A recent study has found that one specific intravenous immune globulin (IVIG) product was more effective than others in counteracting the cognitive deficits, ameliorating b-amyloid (Ab) deposits and tau phosphorylation among mice with Alzheimer disease (AD)
Researchers at UT Southwestern Medical Center have discovered an intracellular mechanism that converts protective intestinal cells into disease-driving pathogenic cells, a finding that could lead to improved treatments for patients with inflammatory bowel disease.
New recommendations from the American Association of Neuromuscular and Electrodiagnostic Medicine advise against using IVIG in some cases.
According to data, patients with autoimmune diseases or long COVID who used a personalized digital care program (DCP) developed by Mymee alongside other nonpharmacologic strategies were more likely to achieve better health-related quality of life scores.
The U.S. Food and Drug Administration (FDA) has converted Leqembi (lecanemab-irmb), indicated to treat adult patients with Alzheimer’s disease, to traditional approval following a determination that a confirmatory trial verified clinical benefit.
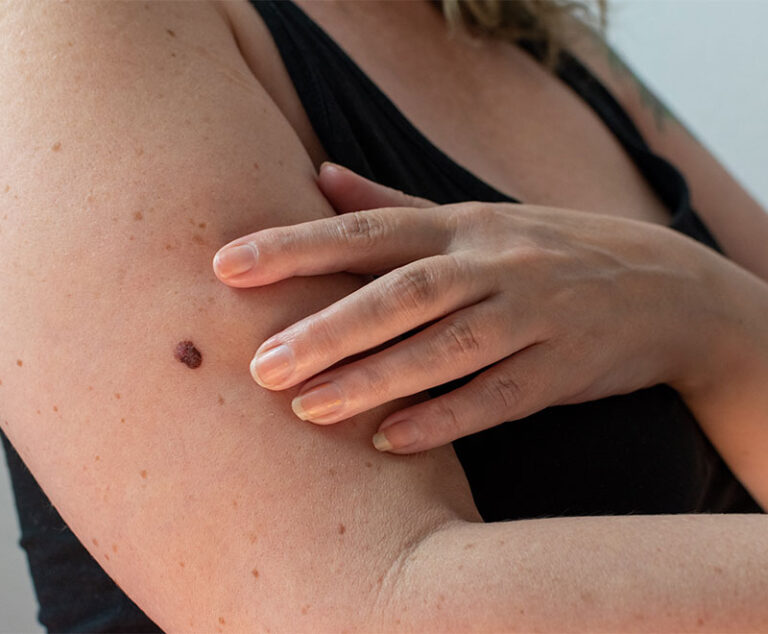
Researchers from Sanford Burnham have found that a protein known to be active in immune cells is also active inside melanoma cells, helping promote tumor growth, suggesting that targeting this protein with new drugs may deliver a powerful double hit to melanoma tumors.
Short-term human albumin infusion significantly reduced the incidence of hyponatremia and increased the rate of improvement of hyponatremia in hospitalization hepatic cirrhosis patients.
A systematic search of leading databases and registries by Chinese collaborators compared the efficacy of IVIG to routine care for hospitalized COVID-19 patients.