FDA Expands Approval to wilate for Prophylaxis in All Types of VWDr
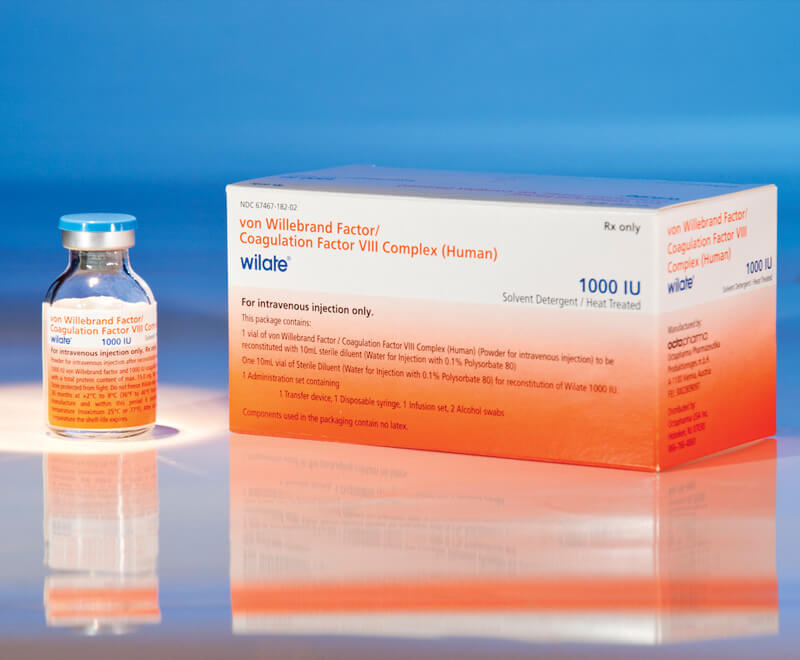
Octapharma USA’s wilate, von Willebrand factor (VWF)/coagulation factor VIII complex (human) lyophilized power for solution for intravenous injection, has been given expanded approval by the U.S. Food and Drug Administration (FDA) for routine prophylaxis to reduce the frequency of bleeding episodes in adults and children aged 6 and older with any type of von Willebrand disease (VWD).
Hyaluronidase-Facilitated Subcutaneous Immune Globulin Effective as Maintenance Therapy for CIDP: Pivotal Trial Results
In a study, researchers found that fSCIG 10% was more effective in preventing CIDP relapse than placebo, supporting its potential use as maintenance CIDP treatment.
Outpatient Albumin Infusions Reduce Hospitalizations in Decompensated Cirrhosis: Retrospective Cohort Study

A team of Australian investigators conducted a retrospective cohort study to determine whether there is a significant benefit in more severely affected cases regularly infused with albumin.
Cancer Drug Shows Promising Results in Clinical Trial

An international early-phase clinical trial has found a “two-for-one” cancer immunotherapy, tebotelimab, is potentially more effective and at least as safe as standard immunotherapies.
FDA Approves Vaccine for Meningococcal Disease in Adolescents
Pfizer’s PENBRAYA, a vaccine for meningococcal groups A, B, C, W and Y, has been approved by the U.S. Food and Drug Administration (FDA).
FDA Approves Celltrion’s Zymfentra to Treat Ulcerative Colitis and Crohn’s Disease
The U.S. Food and Drug Administration (FDA) has approved Celltrion’s Zymfentra, a subcutaneous injection formulation of its infliximab Remsina, for maintenance therapy in adults with moderately to severely active ulcerative colitis and Crohn’s disease following treatment with an infliximab administered intravenously.
FDA Approves Wezlana, First Interchangeable Biosimilar to Stelara
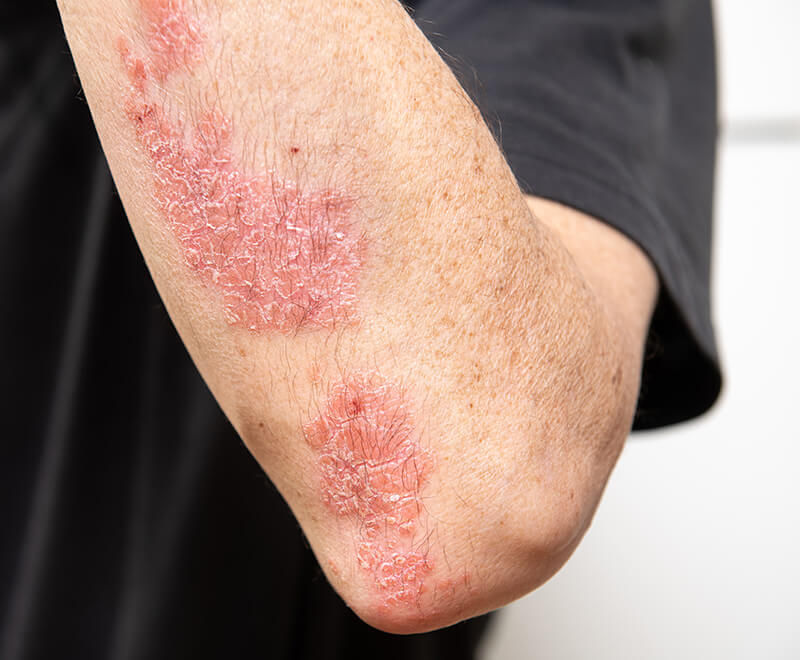
The U.S. Food and Drug Administration (FDA) has approved Wezlana (ustekinumab-auub) as a biosimilar to and interchangeable with Stelara for adult patients with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy; active psoriatic arthritis; moderately to severely active Crohn’s disease; and moderately to severely active ulcerative colitis.
The Proven Success of Immune Globulin Therapy to Treat Chronic Inflammatory Demyelinating Polyneuropathy
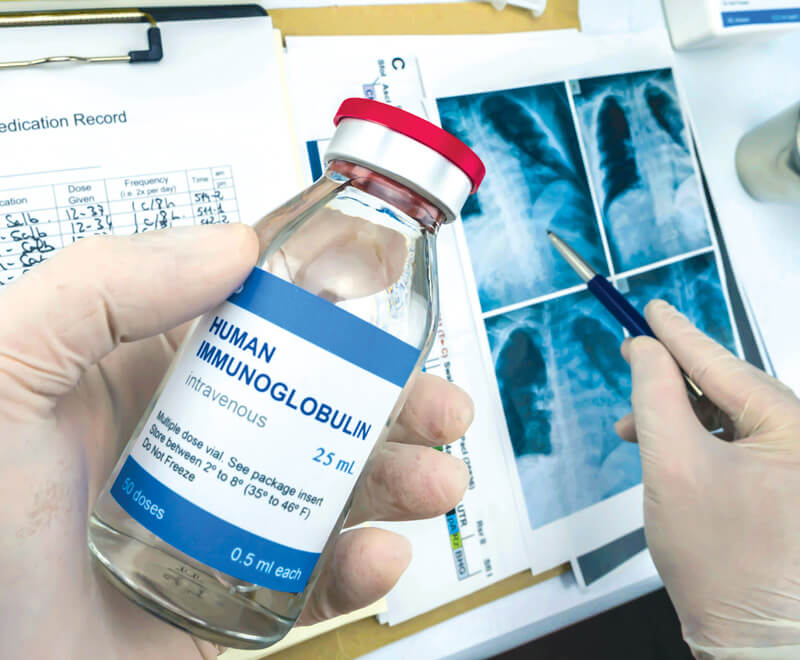
Numerous studies show the efficacy of both IVIG and SCIG for treating CIDP, making these the best treatment options for this rare neurological condition.
Platelet-Rich Plasma Therapy: The Future of Healing?
The effectiveness of this controversial therapy in treating musculoskeletal disorders and other arthritic conditions is uncertain due to mixed results. Nevertheless, the PRP industry is booming.
Coming Soon: Dried Plasma for Hemorrhagic Trauma in the Prehospital Setting

It is now well-established that prehospital transfusion of plasma, with its balanced mix of coagulation proteins, counters the hemodilution and progressive derangement of hemostatic mechanisms caused by crystalloid-based resuscitation, improving the changes of survival in severely bleed trauma victims.