Update on Treating Neutropenia
While neutropenia can be a life-threatening condition, physicians have many tools to treat it.
New Vaccine Could Lower ‘Bad’ Cholesterol by as Much as 30 Percent
A new vaccine currently in development can effectively and affordably lower levels of “bad’ cholesterol in the body, a health problem that affects almost two in five adults in the U.S.
World’s First Gene Therapies Approved to Treat Sickle Cell Disease
The U.S. Food and Drug Administration has approved two treatments, Casgevy and Lyfgenia, representing the first cell-based gene therapies for the treatment of sickle cell disease in patients 12 years and older.
Hizentra Vials to Be Discontinued and Replaced with Prefilled Syringes

CSL Behring is discontinuing all sizes of Hizentra vials in the U.S. by the end of September 2024.
Discovery Could Predict Immunotherapy Response in Melanoma

Researchers have discovered a rare type of immune cell that may predict how likely some patients with skin cancer will respond to immunotherapy treatment.
First Cancer Patients Receive mRNA Therapy in Clinical Trial
Imperial College Healthcare NHS Trust has enrolled the first United Kingdom (UK) patients who have received an experimental messenger RNA (mRNA) therapy — a type of immunotherapy treatment called mRNA-4359 — in its Phase I/II clinical trial.
FDA Expands Approval to wilate for Prophylaxis in All Types of VWDr
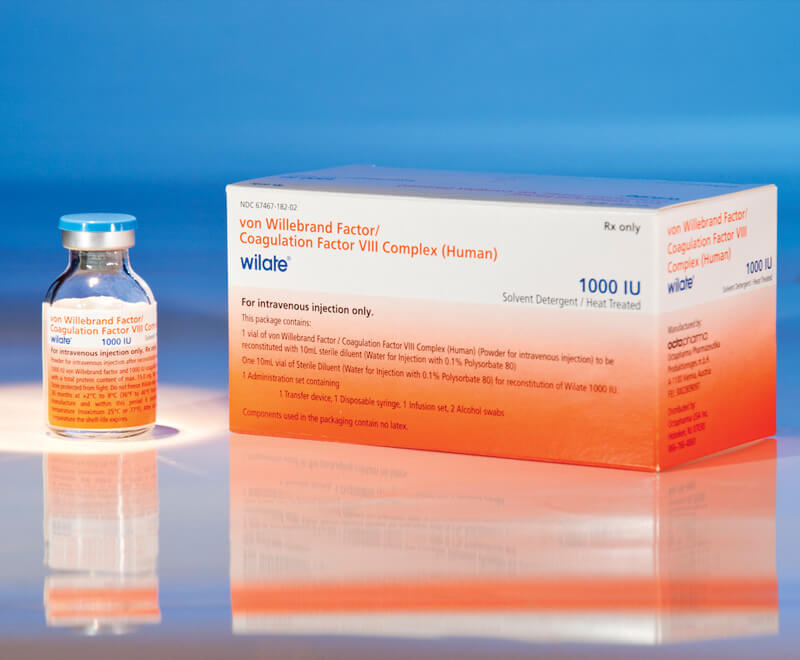
Octapharma USA’s wilate, von Willebrand factor (VWF)/coagulation factor VIII complex (human) lyophilized power for solution for intravenous injection, has been given expanded approval by the U.S. Food and Drug Administration (FDA) for routine prophylaxis to reduce the frequency of bleeding episodes in adults and children aged 6 and older with any type of von Willebrand disease (VWD).
Hyaluronidase-Facilitated Subcutaneous Immune Globulin Effective as Maintenance Therapy for CIDP: Pivotal Trial Results
In a study, researchers found that fSCIG 10% was more effective in preventing CIDP relapse than placebo, supporting its potential use as maintenance CIDP treatment.
Outpatient Albumin Infusions Reduce Hospitalizations in Decompensated Cirrhosis: Retrospective Cohort Study

A team of Australian investigators conducted a retrospective cohort study to determine whether there is a significant benefit in more severely affected cases regularly infused with albumin.
Cancer Drug Shows Promising Results in Clinical Trial

An international early-phase clinical trial has found a “two-for-one” cancer immunotherapy, tebotelimab, is potentially more effective and at least as safe as standard immunotherapies.
