New Assay to Accurately Measure FXI Levels During IVIG Therapy

GC Biopharma has developed a proprietary enzyme-linked immunoassay (ELISA) that effectively mitigates heterophilic antibody interference to precisely measure factor XI (FXI) levels.
Novel Flu Vaccine Offers Broad Protection Against Multiple Flu Viruses

A potentially effective mucosal vaccine platform has been developed to encourage broad, protective immunity against multiple influenza virus infections.
Flu Vaccine Even with Infection Reduces Risk of Heart and Stroke

A new study shows that influenza vaccination may help protect against heart attack and stroke even when it does not prevent people from getting the flu.
High-Dose Flu Vaccine Significantly Lowers Risk of Developing Alzheimer’s

Researchers have found that adults 65 and older who received a high-dose influenza vaccine had a significantly lower risk of developing Alzheimer’s disease compared to those who received the standard dose.
FDA Approves Gene Therapy for Rare Immune Disorder

FDA has approved Kresladi (marnetegragene autotemcel), the first gene therapy for the treatment of severe leukocyte adhesion deficiency type I.
IVIG Effective in Treating Infants with HDFN
A recent study has found intravenous immune globulin (IVIG) therapy in infants with hemolytic disease of the fetus and newborn (HDFN) found that it could be beneficial.
Airborne Spread of Flu Is Reduced by Targeting Two Flu Proteins

A new study demonstrates a way to stop the influenza virus from leaping from one host to the next while continuing to keep the virus from replicating inside the host.
AREXVY RSV Vaccine Approved for Expanded Age Indication
The U.S. Food and Drug Administration has expanded the approved age indication of AREXVY to adults aged 18 to 49 years at increased risk for lower respiratory tract disease caused by RSV.
IVIG Plus IVMP Does Not Increase Remission Rates in CIDP Patients

According to a recent study, adding intravenous methylprednisolone to intravenous immune globulin induction therapy does not significantly increase remission rates among patients with chronic inflammatory demyelinating polyradiculoneuropathy.
New Skin Patch More Accurately Monitors Immune Conditions
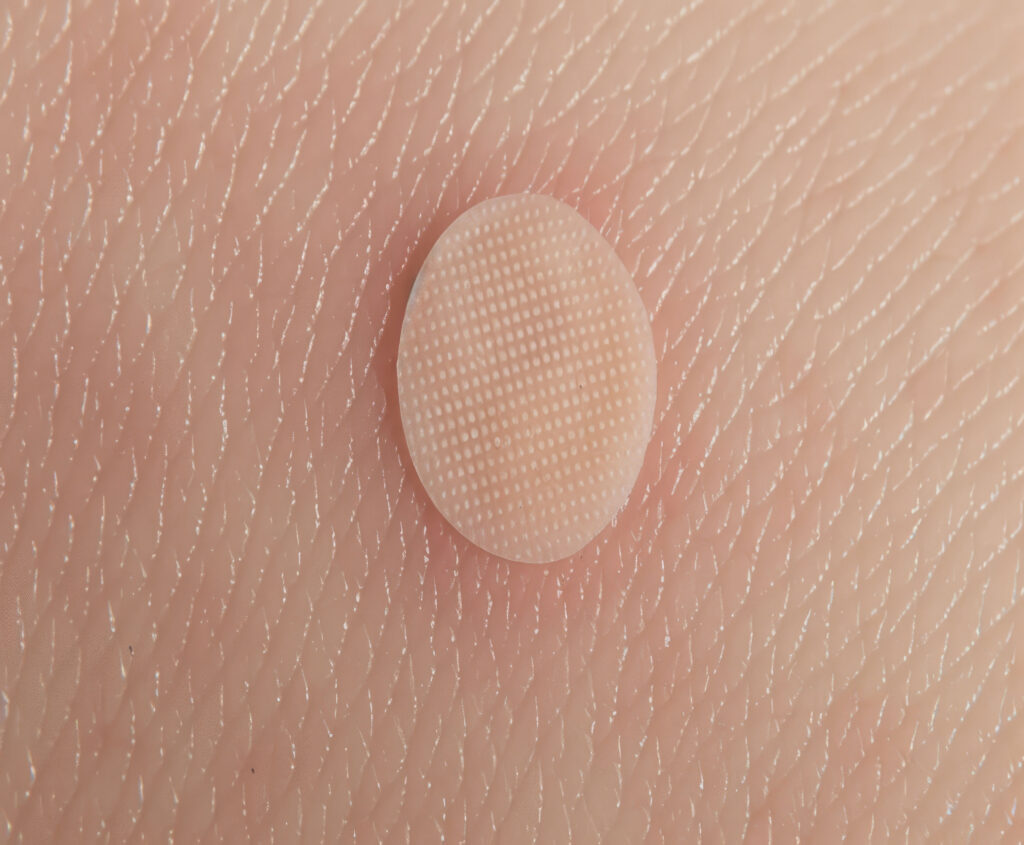
Researchers have developed a bandage-like microneedle patch that samples key cells often missed in routine blood samples and biopsies without the need for taking blood or surgical biopsies.