New and Better Hemophilia A Treatment Options Just Keep Coming
- By Keith Berman, MPH, MBA
ONE CANNOT name a chronic health disorder whose clinical management has undergone a more dramatic evolution in our lifetimes than hemophilia A. Nor could one imagine a single-disease area of biopharmaceutical development with as many major plot twists, the most surprising of which is playing out as you read these words.
How Far We’ve Come
Prior to the introduction of the first human factor VIII (FVIII) concentrates in the late 1960s, patients experiencing a painful joint or soft tissue bleed were usually hospitalized and transfused with large volumes of fresh frozen plasma (FFP) in an effort to deliver just enough FVIII to control the bleed. Inevitably, years of joint bleeds would result in worsening hemarthrosis and progressive physical disability. Replacement of FFP by potent FVIII concentrates purified from pooled units of donor plasma both enabled the administration of much more factor and allowed patients to self-infuse at home, dramatically reducing the time from detection of the bleed to treatment.
In response to the 1980s AIDS crisis that infected and killed thousands of hemophilia patients, new generations of heat-treated, chemically-inactivated, monoclonal antibody-purified and nanofiltered human FVIII concentrates essentially eliminated HIV contamination and transmission risk, while additionally conferring protection against hepatitis B and C virus infection. A decade later, approvals of the first synthetic human FVIII concentrates produced by recombinant DNA techniques provided yet more reassurance of safety for physicians, patients and caregivers traumatized by the AIDS crisis.
These advances in the safety and purity of FVIII concentrates coincided with universal adoption of a game-changing advance in clinical management: regular prophylactic infusions of FVIII to help prevent bleeds from occurring in the first place. Then came the approval of Biogen Idec’s Eloctate in 2014, heralding a new generation of genetically modified longacting recombinant FVIII products whose extended intravascular persistence allows for major reductions in self-infusion frequency, translating into improved patient compliance with their prescribed prophylaxis regimens.
Today, physicians and their patients can choose from 16 FVIII products approved for the treatment of hemophilia A, including six recombinant, six longacting recombinant and four plasma-derived FVIII products.1
Beyond FVIII Replacement: Hemlibra, Humpavzi and Alhemo
As transformative as each of these advancements in replacement therapy with FVIII concentrates have been for the management of hemophilia A, perhaps the most surprising new wave of innovations yet arrived in late 2017 with the approval of Hemlibra, an entirely novel non-factor product developed by Japan’s Chugai Pharmaceutical and manufactured by Genentech, a Roche company. Hemlibra (emicizumab-kxwh) is a modified IgG4 bispecific humanized monoclonal antibody (MAb) that binds factor IXa and factor X, bridging these two key elements of the coagulation pathway to restore the function of the missing FVIII normally required for hemostasis.
Approved for hemophilia A patients of any age both with or without FVIII inhibitors, Hemlibra is specifically intended for routine prophylaxis to prevent or reduce the frequency of bleeding episodes. Not unlike subcutaneous human IgG immune globulin preparations, the pharmacokinetics of this uniquely designed MAb allows for subcutaneous self-administration on a weekly, biweekly or potentially even monthly basis. By comparison, the maximum intertreatment interval for any of the approved long-acting FVIII products is one week or less, and all must be self-administered by intravenous infusion.
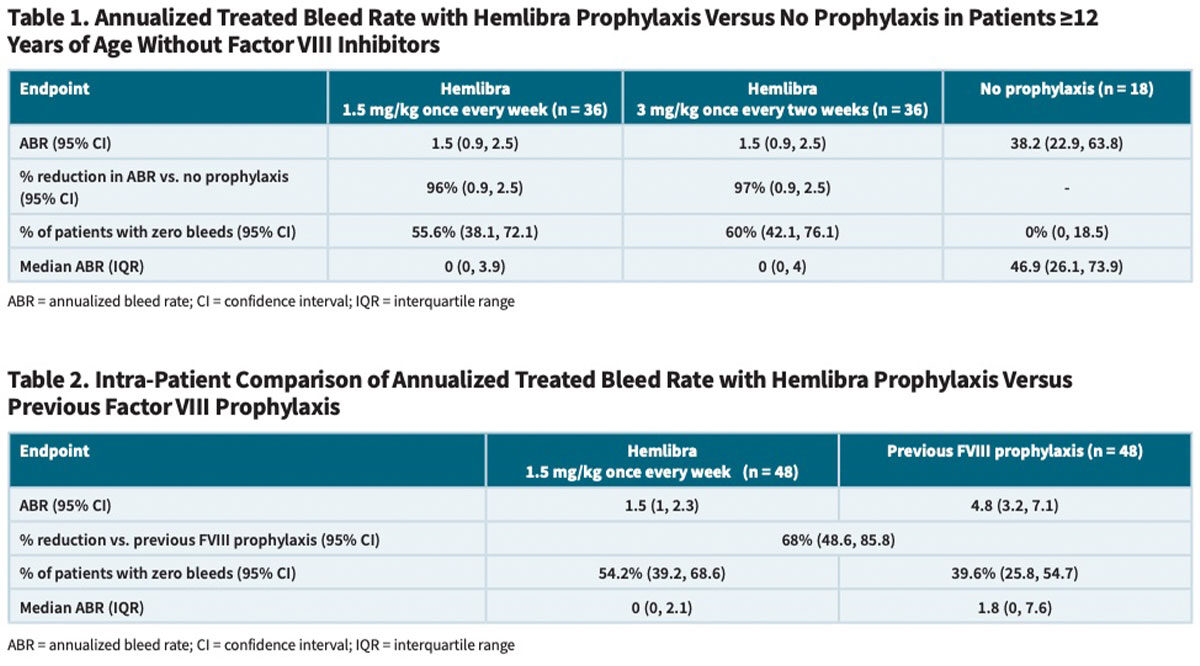
But the benefits of Hemlibra go well beyond its superior convenience and ease of administration. The Roche-sponsored HAVEN 3 study, the largest of several interventional trials documenting its efficacy and safety, evaluated Hemlibra in a total of 152 adult and adolescent males previously self-treated prophylactically or on-demand with FVIII concentrates in response to bleeding events. While many more outcome measures were reported (e.g., treated spontaneous bleeds and joint bleeds), tables 1 and 2 summarize 1) the overall performance of weekly or biweekly Hemlibra against concurrent episodic (on-demand) FVIII treatment and 2) weekly Hemlibra against previous FVIII prophylaxis in the same individuals.2
Each hemophilia A patient’s status and needs are different, and many can continue to be successfully managed with factor therapy. But Hemlibra’s strong overall comparative efficacy findings and its simplicity of use have enabled it to capture a very substantial share of the U.S. and EU5* hemophilia A patient market, with well over 20,000 patients treated with Hemlibra worldwide.3
Hympavzi (marstacimab-hncq). Last October, a second entirely novel nonfactor treatment, Pfizer’s Hympavzi, was approved by the U.S. Food and Drug Administration (FDA) for routine prophylaxis to prevent or reduce the frequency of bleeding episodes in adults and pediatric patients 12 years of age and older with hemophilia A or B without inhibitors.
This IgG1 antibody targets a specific domain of the tissue factor pathway inhibitor (TFPI), which negatively regulates thrombin generation within the extrinsic coagulation pathway by inactivating the protease functions of factors Xa, VIIa and the tissue factor complex. Hympavzi’s action occurs downstream from FVIII or factor IX, so it is similarly effective in hemophilia B, as well as hemophilia A patients. But like Hemlibra, Hympavzi is easy to selfadminister (on a weekly basis only) with a quick subcutaneous injection.
Compared to prior on-demand factorbased therapy, Hympavzi prophylaxis reduced treated bleeds in 33 subjects enrolled in the BASIS study by more than 90 percent, from 38 to just over three bleeds. In a larger cohort of 83 subjects with hemophilia A and B on prior routine factor-based prophylaxis, Hympavzi prophylaxis significantly reduced the annualized bleeding rate (ABR) from nearly eight bleeds to five bleeds.
Alhemo (concizumab-mtci). Last December, just two months after it approved Hympavzi, FDA granted marketing approval for Novo Nordisk’s Alhemo, a second proprietary MAb-based TFPI antagonist. Launched in February, Alhemo is specifically indicated as oncedaily prophylaxis to prevent or reduce the frequency of bleeding episodes in adults and pediatric patients 12 years of age and older with hemophilia A or B with inhibitors. Alhemo can be dosed subcutaneously by the patient using a prefilled pen and fine gauge needle.
The estimated ABR for hemophilia A and B patients with inhibitors on Alhemo for at least 32 weeks was 1.7 bleeds, a reduction of 86 percent relative to the ABR of 11.8 bleeds for patients whose bleeds were conventionally managed with a bypassing agent (eptacog alfa; NovoSeven).4
Alhemo “provides a much-needed alternative to the current standard of care in hemophilia B with inhibitors, while offering patients with hemophilia A with inhibitors more treatment options, ultimately providing more patients with inhibitors the opportunity to personalize their care and address current treatment gaps,” said Indiana Hemophilia & Thrombosis Center CEO and co-medical director Amy Shapiro, MD.
Following behind Hemlibra, Hympavzi and Alhemo are several other very promising non-factor treatments, including two in particular that are currently in late-stage clinical development:
Mim8 (Novo Nordisk). This subcutaneously administered factor VIIIa mimetic bispecific antibody bridges factor IXa and X together upon activation, thereby replacing missing FVIII. In clinical trial subjects with hemophilia A aged 12 years and older with or without inhibitors, those given Mim8 once weekly and once monthly, respectively experienced 97 percent and 99 percent reductions in treated bleeds compared to those who received no prophylaxis treatment. Remarkably, 86 percent and 95 percent of subjects, respectively, treated with once-weekly and once-monthly Mim8 experienced no treated bleeds over the 26-week treatment period.
In an intra-patient analysis of subjects previously managed with FVIII prophylaxis, once-weekly and oncemonthly Mim8 demonstrated reductions of 48 percent and 43 percent in treated bleeds, respectively; again, roughly twothirds of these study subjects experienced no treated bleeds.
Fitusiran (ALN-AT3) (Sanofi). This investigational RNA interference (RNAi) agent suppresses the production of antithrombin by targeting the messenger RNA encoded by the SERPINC1 gene that encodes this key regulator of coagulation function. By lowering circulating antithrombin levels, fitusiran administration increases thrombin generation, enhancing hemostasis.5
In a multinational Phase III trial of once-monthly prophylactic subcutaneous injections of fitusiran, patients aged 12 years and older with hemophilia A or B with or without inhibitors were crossed over from their routine use of prophylaxis with bypassing agents or clotting factor concentrates (BPAs/CFCs) to fitusiran prophylaxis for seven months. The observed ABRs were 6.5 while on BPA/CFC therapy versus zero during the fitusiran efficacy period. Estimated mean ABRs were reduced with fitusiran treatment by 79.7 percent and 46.4 percent versus BPA and CFC prophylaxis, respectively. Forty-one of 65 study participants (63.1 percent) experienced zero bleeds with fitusiran versus 11 (16.9 percent) with BPAs/CFCs.
FDA’s decision on Sanofi’s pending application for marketing approval was scheduled for March 28 of this year, so as you read this, fitusiran may already be the fourth new non-factor product approved for the treatment of hemophilia A.
The (Current) Fate of Gene Therapy
Over the last four decades, countless commentaries and media stories have touted gene therapy as the holy grail of severe hemophilia A therapy: a potentially curative one-time treatment that could empower the patient’s own liver cells to produce sufficient sustained titers of functional FVIII, thus providing extended or even lifelong protection against uncontrolled bleeds.
The once far-off dream of gene therapy to cure hemophilia A began in 1984 when Genentech scientists reported they had fully characterized the massive human FVIII gene and expressed active human FVIII in cultured mammalian cells from recombinant DNA clones encoding the complete FVIII gene.6,7
BioMarin Pharmaceuticals was first to market in mid-2023 with Roctavian, an adeno-associated virus vector-based gene therapy for the treatment of adults with severe hemophilia A. The mean ABR was reduced from 5.4 bleeds at baseline to 2.6 bleeds a median of three years after Roctavian administration. Of 112 clinical trial subjects rolled over from FVIII prophylaxis, just five (four percent) did not respond but 17 (15 percent) lost response to Roctavian treatment over a median period of just over two years. In 22 other subjects followed for a longer period, six (27 percent) lost response to Roctavian treatment over a median of 3.6 years.8
But for the large majority of patients who received this one-time gene therapy in clinical trials, it has largely or entirely freed them from the requirement to selfinfuse factor or any other treatment to restore normal coagulation function. No instances of thromboembolic events or cancers have been observed in trial subjects or patients given Roctavian since approval.**
Why, then, have only a handful of non-trial hemophilia A patients been dosed with Roctavian as the product approaches two years on the market? And why did Pfizer, which has invested heavily in this space, decide last December to pull out of a late-stage R & D collaboration to develop its own hemophilia A gene therapy (giroctocogene fitelparvovec), and then decide in February of this year to discontinue commercialization of Beqvez, its hemophilia B gene therapy approved just a year ago?
“[Roctavian is] extremely expensive. And I think that it’s going to take some time to be able to convince payers to cover this product,” said Roctavian clinical study investigator Steven W. Pipe, MD, in an interview last year. “And then secondly… there’s a lot of infrastructure that needs to be built out at the institutional level, in order to be a gene therapy infusion center.”9 These patient access-related issues continue to adversely impact usage, and unanswered questions remain concerning the durability and potential long-term side effects of hemophilia gene therapy.
But another consideration cannot be ignored: Prophylaxis treatment options available today have all but eliminated the fear of serious bleeds for many hemophilia A patients, and non-factor treatments in particular have made managing their condition far easier than ever before. “Unlike other genetic disorders for which gene therapy is available, hemophilia A and B each have effective treatment options that have more long-term safety and efficacy data,” a team of clinicians at the University of North Carolina at Chapel Hill recently observed. “Given available alternative treatment options…a minority of patients are eager to undergo gene therapy treatment at present, and many are pleased with their current treatment regimen.”10 Put another way, the risk-benefit argument for gene therapy — at least at the moment — doesn’t appear to add up for many patients or physicians upon whom they rely for information and advice.
The Dream of a Normal Life with Hemophilia A
Like few other areas of drug development, hemophilia A new product research has married cutting-edge genetic and protein engineering and rigorous clinical science to produce wave after wave of new and better treatment options. The sparse adoption of Roctavian and recent abandonment of clinical-stage gene therapies is obviously disappointing for their developers, but at least in part this reflects the larger success of an astonishing output of new therapies with unprecedented efficacy and convenience.
Thanks to these new products that have and continue to emerge from industry research laboratories, patients and providers today must search their memories or history books to recall that people with severe hemophilia A were once consigned to a lifetime of painful emergent bleeds, severely debilitating joint disease and progressive physical limitations. The dream of a normal life for persons with hemophilia A has arrived.
* European Union Five (France, Germany, Italy, Spain and the United Kingdom)
** FDA does caution that Roctavian’s viral vector-introduced B-domain deleted FVIII gene may still carry the theoretical risk of hepatocellular carcinoma.
References
- FFF Enterprises. Coagulation Products Reference Catalog (9/25/2024). Accessed at www.fffenterprises.com/assets/downloads/FFF_ReferenceChart-Coagulation.pdf.
- HEMLIBRA (emicizumab-kxwh) injection, for subcutaneous use. Full prescribing information. Revised 01/2024.
- Genentech. Number of people with hemophilia A treated with HEMLIBRA as of April 2023. Accessed at www.hemlibra.com.
- Alhemo (concizumab-mtci) injection, for subcutaneous use. Full prescribing information. Revised 12/2024.
- Pasi KJ, Rangarajan S, Georgiev P, et al. Targeting of antithrombin in hemophilia A or B with RNAi therapy. New Engl J Med 2017;377:819-28.
- Gitschier J, Wood WI, Goralka TM, et al. Characterization of the human factor VIII gene. Nature Nov;312(5992):326-30.
- Wood WI, Capon DJ, Simonsen CC, et al. Expression of active human factor VIII from recombinant DNA clones. Nature 1984 Nov;312(5992):330-7.
- ROCTAVIAN (valoctocogene roxaparvovec-rvox) suspension for intravenous infusion. Full prescribing information. Revised 06/2023.
- Johnson V. A slow embrace: Hemophilia’s gradual adoption of gene therapy. CGTlive. April 17, 2024. Accessed at www.cgtlive.com/view/hemophilia-gradual-adoption-gene-therapy.
- Lee K, Berkowitz C, Jackson K, et al. Challenges in establishing best practices for gene therapy informed consent for hemophilia A/B. Genetics in Medicine 2024; 2(Suppl 1):101142.