Myths & Facts: GLP-1 Drugs for Weight Loss
While there has been a surge in GLP-1 weight-loss drugs since first being approved a decade ago, misconceptions remain that could lead to many patients failing to achieve their weight-loss goals.
- By Ronale Tucker Rhodes, MS
WEIGHT LOSS is one of the most talked-about topics. The reason? The number of obese individuals in the U.S. is growing at an alarming rate. Obesity is defined as a body mass index higher than 30 percent. In 1980, the obesity prevalence was 15 percent. This progressed to an obesity prevalence of 31 percent in 2000 and then 42 percent in 2020. And, while there is no consensus on the cause of this growing obesity rate — reduction in physical activity, increase in dietary fat, increase in sugar and sugary beverages, increase in ultra-processed foods, higher cost of healthier diets — there’s no denying that Americans need solutions.
It’s no wonder, then, that when talking about weight loss today, discussion is dominated by the new GLP-1 (glucagon-like peptide-1) drugs. But, this “new drug on the block” really isn’t so new. Researchers began searching for insulin stimulating factors about 100 years ago. And, that’s what GLP-1 drugs are: a type of peptide hormone that can stimulate the pancreas to produce more insulin after meals, as well as slow the movement of food from the stomach into the small intestine to help suppress appetite. Despite years of research, the first discovery of a GLP-1 wasn’t until 1984, and upon further research into its applications, a drug was approved by the U.S. Food and Drug Administration (FDA) to treat type 2 diabetes. After that, clinical studies and post-release surveillance observations showed that individuals prescribed the drug were also losing weight. So, pharmaceutical companies started investigating the use of GLP-1s for weight-management purposes, and the first GLP-1 weight-loss drug, Saxenda (liraglutide), was approved by FDA in 2014 (it was previously approved to treat diabetes under the name Victoza).
Today, there are three GLP-1 weight-loss drugs available: Saxenda; Wegovy (semaglutide), which was approved in 2021 (it was previously available for diabetes as Ozempic and in an oral form for diabetes as Rybelsus); and Zepbound (tirzepatide), which was approved in 2023 (it was previously available to treat diabetes under the brand name Mounjaro). Zepbound is the first GLP-1/GIP (gastric inhibitory polypeptide) medication, which is thought to be more potent than GLP-1 alone.
Between 2014 and now, prescriptions for these GLP-1 drugs have increased a whopping 300 percent. Yet, many myths surround who should be prescribed these drugs, how they work and what they can and cannot do.1
Separating Myth from Fact
Myth: Anyone can take GLP-1 weight-loss drugs.
Fact: GLP-1 weight-loss drugs are typically prescribed to people with a BMI of 30 or higher.2 Both Saxenda and Wegovy can be prescribed to individuals 12 years and older, while Zepbound can be prescribed only to adults. However, they are not for everyone. People with a history of medullary thyroid cancer, gallbladder disease or pancreatitis, as well as those with multiple endocrine neoplasia syndrome type 2 should avoid taking these drugs.2
In fact, according to Jaime Almandoz, MD, while more than 70 percent of U.S. adults are overweight or obese, only four percent of these people are prescribed GLP-1 weight-loss medicines due to concerns about safety or effectiveness, lack of healthcare provider training in weight management, excessive costs due to insurance companies refusing coverage and a toxic stigma that portrays obesity as a lack of willpower.3
There are also strict guidelines for who qualifies for these medications, and there’s a spectrum of little-talked-about weight-loss medications available on the market that aren’t GLP-1s.4
Myth: All GLP-1 weight-loss drugs are the same.
Fact: No, they are similar, but not the same. Saxenda, Wegovy and Zepbound are all GLP-1 receptor agonists that work by stimulating the GLP-1 receptor in the brain to help people feel full.5
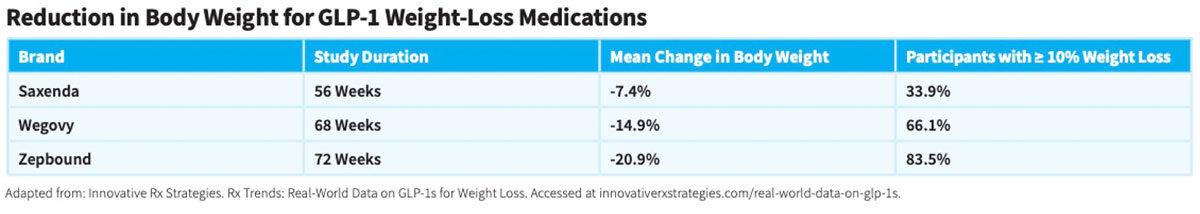
Saxenda (liraglutide) is injected subcutaneously (under the skin) usually once daily, with or without meals. It is started at a lower dose and then gradually increased over the weeks. In a large 56-week clinical trial that evaluated liraglutide in individuals who were overweight or had obesity, as well as had diabetes or prediabetes, participants reported statistically significant weight loss compared with those who took the placebo at the end of the study. Sixty-three percent of the participants lost at least five percent of their body weight and about 33 percent lost at least 10 percent of their body weight.6 In another study of people without diabetes, Saxenda was more effective for weight loss than lifestyle interventions alone, and weight loss was sustained for more than two years.7
Wegovy (semaglutide) is injected under the skin once weekly, at the same time every week, with or without meals. In studies, semaglutide was more effective at reducing body weight and HbA1c than placebo. When used in addition to lifestyle interventions, 2.4 mg of semaglutide once weekly resulted in an average weight loss of 15 percent. More people who took semaglutide (86 percent) lost at least five percent of their body weight compared with those on placebo (13 percent). And about 70 percent of users lost at least 10 percent of their body weight.8 Studies have also shown semaglutide to be superior to some other weight-loss agents. Compared with liraglutide, more semaglutide users achieved weight loss greater than five or 10 percent.9
Zepbound (terzepatide) is also injected subcutaneously once weekly at a recommended dosage of 5 to 15 mg. Two large 72-week clinical trials that evaluated terzepatide in individuals who were overweight or obese found that by the end of the study, people who also dieted and exercised reported statistically significant weight loss compared to those who took the placebo.10
Myth: All GLP-1 agonists work the same for all people.
Fact: The drugs’ efficacy varies from person to person. According to Laura Buford, MD, “factors such as genetics, lifestyle and underlying health conditions all play a role in how an individual responds to these treatments. For example, some people may experience more significant weight loss, while others may see more modest results.” What’s more, diet, exercise and adherence to the treatment plan affect the outcomes. And, given the individual variability, Dr. Buford says it’s necessary for healthcare providers to personalize the plan for each individual such as adjusting the dosage, combining treatments or incorporating lifestyle modifications to optimize results for each person.11
Myth: GLP-1 weight-loss drugs are just another diet pill.
Fact: Actually, GLP-1 weight-loss drugs take a completely different approach than diet pills. Diet pills focus on suppressing appetite or increasing metabolism, whereas GLP-1 agonists work by mimicking the action of the GLP-1 hormone that is naturally produced in the body and plays a crucial role in regulating appetite and blood sugar levels. According to Dr. Buford, “by enhancing the effects of GLP1, these medications help to reduce hunger and increase feelings of fullness, leading to reduced caloric intake without the need for extreme dieting or willpower. This mechanism of action is more aligned with the body’s natural processes, making it a more sustainable option for weight loss.”11
Myth: Using compounded GLP-1 drugs is just as safe as using brand-name GLP-1 medicines.
Fact: According to FDA, “a compounded drug might be appropriate if a patient’s medical need cannot be met by an FDA-approved drug, or the FDA-approved drug is not commercially available. However, compounded drugs are not FDA approved,” which means FDA does not review them for safety, effectiveness or quality before they are marketed. In fact, FDA has identified some areas of concern for compounded GLP-1 drugs and is working with its state regulatory partners regarding them. Concerns include reports of adverse events, some requiring hospitalization, that may be related to dosing errors or doses beyond what is in the FDA-approved drug label, which means using more product in a single dose, taking doses more frequently or increasing the amount more quickly (titration schedule).12
Myth: It’s OK to purchase GLP-1 drugs from online or compounding pharmacies that don’t require a prescription.
Fact: It’s unwise to purchase GLP-1 medications from anywhere other than major retailers who require a prescription, because individuals may actually be getting a counterfeit drug. In fact, counterfeit Ozempic sales are on the rise, and these drugs could contain the wrong ingredients; contain too little, too much or no active ingredient at all or other harmful ingredients; and are illegal.12
To spot whether Ozempic is counterfeit, FDA encourages patients and healthcare providers to double-check Ozempic boxes, which might have spelling errors, and the batch number on the box may not match the product strength stated on the same box and pen. The label on a counterfeit pen could also have poorer quality, and it may not stick well to the pen. A genuine Ozempic pen also does not extend in length when you try to set the dose with the dial.13 In addition, the World Health Organization advises not to distribute, use or sell products labeled with batch numbers listed in its Annex.14,15
Myth: The GLP-1 injections are painful.
Fact: In general, the injections aren’t painful because the injection pen uses a tiny needle that is inserted just under the skin. These subcutaneous injections are significantly less painful than injections into muscles such as flu shots. One study showed that injections of semaglutide (the active ingredient in Wegovy), which is administered for diabetes or weight loss in the skin of the abdomen, thigh or upper arm, produced little to no injection‐site pain.16
However, there have been reports of injection site discomfort from those new to injecting GLP-1s. But, this side effect can be avoided by following a few best practices before, during and after the injections. These include injecting just under the skin’s surface in the stomach, upper arm or thigh, ideally into fat; not injecting into veins or muscles; rotating injection sites and changing the site for each dose to help prevent skin irritation and scar tissue buildup; staying at least two inches away from the belly button when injecting into the stomach; not injecting into areas that are red, bruised or tender; and avoiding injecting into areas with stretch marks or scars.17
Myth: People don’t have to exercise or eat healthfully when taking GLP-1 drugs.
Fact: According to the labels on these drugs, people do have to exercise and eat healthfully while using them. Specifically, the label states these medications should be used in combination with a “reduced calorie diet and increased physical activity.” HaVy Ngo-Hamilton, PharmD, pharmacist and clinical consultant for BuzzRx, states that “the amount of diet and exercise will vary among users, leading to various degrees of weight loss. However, consistent physical activity is vital to achieve and maintain the desired weight loss.”
Following a balanced diet is important when taking GLP-1 medications because it can help improve the effectiveness of the medication in managing blood sugar levels and weight. “Because these medications trigger insulin production, slow the emptying of the stomach and cause you to feel full faster and for longer periods of time, eating unhealthy foods or larger portions of food may cause negative gastrointestinal side effects, including abdominal pain, nausea or vomiting,” Ngo-Hamilton says.18
Myth: GLP-1 drugs are unsafe with numerous side effects.
Fact: Actually, these drugs are very safe as proven by extensive clinical testing. Results of these trials showed “GLP-1 agonists have a favorable safety profile, with most side effects being mild and temporary. Common side effects include nausea, diarrhea and constipation, which typically subside as the body adjusts to the medication.” There are strategies to manage and minimize side effects. For instance, individuals can start with a lower dose that is gradually increased to reduce the severity of side effects. And, taking medication with food or adjusting the timing of doses can help.11
Myth: GLP-1 drugs cause suicidal ideation.
Fact: In July 2023, the European Medicines Agency began reviewing reports of patients causing self-harm or having suicidal ideation after taking weight-loss drugs that contain semaglutide. However, a meta-analysis conducted at Case Western University in collaboration with the National Institutes of Health and FDA demonstrated no link between anti-obesity drugs and increased suicidal ideation.19
In the study, the researchers used a large electronic health record database to conduct a nationwide retrospective cohort study to assess the association of semaglutide with the incidence and recurrence of suicidal ideation compared with non-GLP1R agonist anti-obesity medications in individuals with overweight or obesity. What’s more, they replicated the same analyses in a separate cohort of patients with type 2 diabetes mellitus by comparing semaglutide with non-GLP1R agonist anti-diabetes medications. According to the researchers, “Contrary to reports of increases in suicidal ideation with semaglutide, our analyses revealed a lower risk for both incidence and recurrence of suicidal ideation in patients prescribed semaglutide compared with non-GLP1R agonist anti-obesity and anti-diabetes medications.”20
“If you look at the study a little bit deeper … there was a decreased risk in [suicidal ideation] in women, men and along every racial group,” said Catherine Varney, DO, obesity medicine director for UVA Health and an assistant professor of family medicine at the University of Virginia. “What we’re seeing is improvements in mental health and metabolic health. This is really exciting for us to see just further evidence of the benefits of these medications beyond weight loss.”19
Myth: Losing weight is the biggest benefit of GLP-1 medicines.
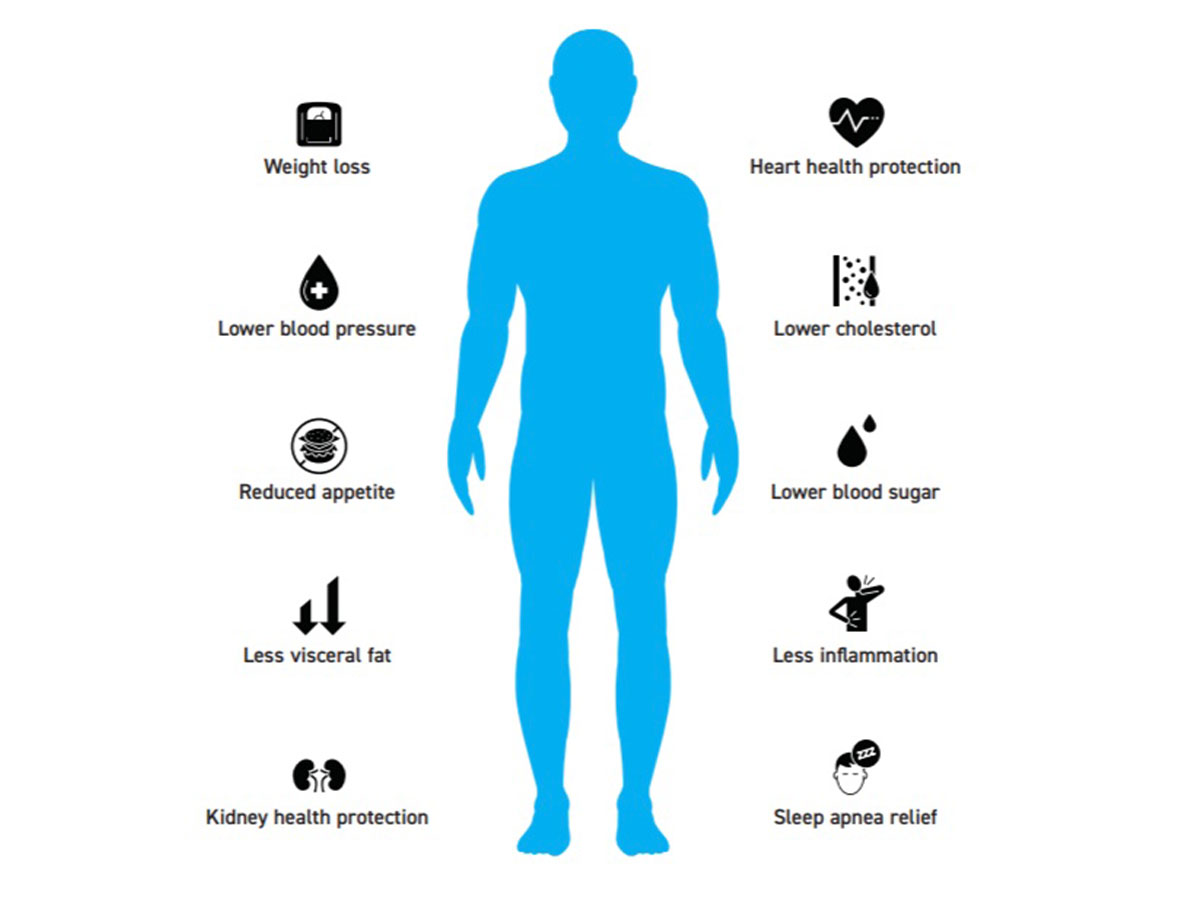
Fact: The benefits of weight loss extend far beyond the scale. Researchers report that GLP-1 drugs prescribed for weight loss may improve cognition and lower dementia and cardiovascular disease risk. Scientists at the Washington Universal School of Medicine in St. Louis and the Veterans Affairs St. Louis Health Care System reviewed the health records of two million veterans who were treated for diabetes from October 2017 through December 2023 to determine the GLP-1 medications’ effects on the body’s organ systems. Some of the veterans took GLP-1 medications, while others were given more traditional drugs sold under brand names such as Jardiance, Glipizide and Januvia. They found widespread associations with the GLP-1 drugs and improvements to cognitive and behavioral health, with benefits including reduced risks of seizures, as well as a lower risk to addiction to substances such as alcohol, cannabis, stimulants and opioids. In addition, the researchers said those taking the GLP-1 drugs also experienced decreased risks of suicidal ideation, self-harm, bulimia and psychotic disorders such as schizophrenia. Their findings also showed a lower risk of neurocognitive disorders such as Alzheimer’s and dementia. However, they noted these benefits were a somewhat modest 10 to 20 percent reduction in most risks.21
Myth: Taking GLP-1 drugs guarantees weight loss.
Fact: While taking GLP-1 drugs may seem like a guaranteed way to reach weight-loss goals, research shows they don’t work for everyone. One clinical trial indicated only about 86 percent of the more than 800 participants achieved “clinically significant” weight loss after taking semaglutide, sold under the brands Ozempic and Wegovy, for 68 weeks, which means a portion of users did not. “Each person responds differently to medications, and the degree of weight loss achieved with treatment can vary based on personal factors,” said Priya Jaisinghani, MD, a clinical assistant professor specializing in obesity medicine at the NYU Grossman School of Medicine.
Of course, there may be factors influencing weight-loss outcomes. First, the dose may not be high enough. A recent study involving 3,390 people taking semaglutide or liraglutide found participants who took high doses of their drug — 1.7 mg, 2.0 mg or 2.4 mg of semaglutide, or 3 mg of liraglutide — lost more weight after a year compared to people taking lower doses. “Increasing the dose of a GLP-1 can sometimes enhance weight loss or maintain consistent weight loss,” explained Fatima Cody Stanford, MD, an obesity medicine scientist at Massachusetts General Hospital Digestive Healthcare Center in Boston. “But this is not guaranteed.”
Physical factors can also play a role. The same study found that people with a higher body mass index lost more weight after a year. Other barriers to weight loss include lifestyle habits, such as not exercising or routinely eating sugary or fatty foods, and medication adherence. According to Dr. Cody Stanford, the drugs are only effective if taken consistently, and people who pause the medication may gain some weight back. GLP-1s can also cause gastrointestinal issues such as nausea and vomiting, which may cause some people to discontinue use due to side effects before achieving their weight-loss goals.22
Myth: Insurance will cover the cost of GLP-1 medicines for people who are prime candidates.
Fact: Unfortunately, this is not always the case. Cost is a barrier for some individuals who would like to go on GLP-1 medications since, according to the Kaiser Family Foundation and the Institute for Clinical and Economic Review, they can cost $1,300 or more for a month’s supply. And, not all insurance will help cover it. For instance, some employers choose to exclude GLP-1 drugs from employer-sponsored health plans, some insurance plans require prior authorization before approval, and Medicare does not cover GLP-1s for weight loss. It is up to each insurer and its chief medical officer to decide if these medications will be covered. While drug manufacturers sometimes offer discount programs, consumers are often left paying hundreds of dollars out of pocket for their prescriptions.23
In addition, a provider may prescribe a GLP-1 off label for obesity when that GLP-1 is covered by insurance only if the person has a type 2 diabetes diagnosis. “Insurance coverage will vary by person and by insurance policy, and recently, more insurance plans have opted not to cover the cost of GLP-1 agonists, particularly those that are being prescribed for off-label use,” Ngo-Hamilton says. “For example, when someone doesn’t have type 2 diabetes, their healthcare provider prescribes Ozempic as off-label use for weight management.”18
Dispelling the Myths Now
While not new, GLP-1 drugs have been approved by FDA only in the last decade to treat obesity. However, they are not miracle drugs; diet and exercise are still important factors for sustainable weight loss while taking a GLP-1 medication. In addition, they are costly medications, they are not always covered by insurance, and they generally need to be taken forever. While some individuals don’t see results, with a better understanding of the facts about these drugs, physicians can help their patients succeed in their weight-loss goals.
References
- Rx History: The Rise of GLP-1s. Innovative Rx Strategies, Jan. 25, 2024. Accessed at innovativerxstrategies.com/rx-history-glp1s.
- Surampudi, V. Semaglutide for Weight Loss — What You Need to Know. UCLA Health, Jan. 12, 2023. Accessed at www.uclahealth.org/news/article/semaglutide-weight-loss-what-you-need-know.
- Almandoz, J. Anti-Obesity Drugs Are Closing the Gap Between Dieting and Bariatric Surgery. UT Southwestern Medical Center, Sept. 7, 2023. Accessed at utswmed.org/medblog/obesitymedication-weight-program.
- Church, T. 5 Must-Know Myths About Weight-Loss Medications. Wondr Health, Jan. 6, 2025. Accessed at wondrhealth.com/blog/5-must-know-myths-about-weight-loss-medications.
- Nguyen, PA. 8 FDA-Approved Drugs for Weight Management. Very Well Health, Feb. 15, 2024. Accessed at www.verywellhealth.com/7-fda-approved-drugs-for-weight-management-7568596.
- Pi-Sunyer, X, Astrup, A, Fujioka, K, et al. A Randomized, Controlled Trial of 3.0 mg of Liraglutide in Weight Management. New England Journal of Medicine, 2015;373(1):11-22. Accessed at pubmed.ncbi.nlm.nih.gov/26132939.
- Astrup, A, Carraro, R, Finer, N, et al. Safety, Tolerability and Sustained Weight Loss Over 2 Years with the Once Daily Human GLP-1 Analog, Liraglutide. International Journal of Obesity (London), 2012;36(6):843-854. Accessed at pubmed.ncbi.nlm.nih.gov/21844879.
- Wilding, JPH, Batterham, RL, Calanna, S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England of Journal Medicine, 2021;384(11):989-1002. Accessed at pubmed.ncbi.nlm.nih.gov/33567185.
- Aroda, VR, Bain, SC, Cariou, B, et al. Efficacy and Safety of OnceWeekly Semaglutide Versus Once-Daily Insulin Glargine as Add On to Metformin (with or without Sulfonylureas) in Insulin-Naive Patients with Type 2 Diabetes (SUSTAIN 4): A Randomised, OpenLabel, Parallel-Group, Multicentre, Multinational, Phase 3a Trial. The Lancet Diabetes & Endocrinology, 2017;5(5):355-366. Accessed at pubmed.ncbi.nlm.nih.gov/28344112.
- Jastreboff, AM, Aronne, LJ, Ahmad, NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine, 2022;387(3):205-216. Accessed at www.nejm.org/doi/full/10.1056/NEJMoa2206038.
- Buford, L. Debunking GLP-1 Myths: The Truth About Medical Weight Loss. Westlake Dermatology, Sept. 24, 2024. Accessed at www.westlakedermatology.com/blog/glp-1-weight-loss-myths.
- U.S. Food and Drug Administration. FDA’s Concerns with Unapproved GLP-1 Drugs Used for Weight Loss, updated Dec. 18, 2024. Accessed at www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/fdas-concerns-unapprovedglp-1-drugs-used-weight-loss.
- Brown, S. How to Spot Fake Ozempic. Very Well Health, Feb. 1, 2024. Accessed at www.verywellhealth.com/fake-ozempic-8553313.
- World Health Organization. Medical Product Alert N°2/2024: Falsified OZEMPIC (semaglutide), June 19, 2024. Accessed at www.who.int/news/item/19-06-2024-medical-product-alert-n-2-2024–falsifiedozempic-(semaglutide).
- World Health Organization. Medical Product Alert No. 2/2024Falsified OZEMPIC (semaglutide) Identified in the WHO Regions of Americas and Europe. Accessed at cdn.who.int/media/docs/default-source/ substandard-and-falsified/correction-n2_2024_ozempic_ en.pdf?sfvrsn=17d87e2d_12.
- Gussone, F. Do Wegovy Injections Hurt? Accessed at ro.co/weightloss/does-wegovy-injection-hurt.
- Crider, C. Where Do You Inject Semaglutide? Healthline, updated July 26, 2023. Accessed at www.healthline.com/health/where-do-youinject-semaglutide.
- Mayer, A. 8 Common Myths About Weight Loss Drugs Like Wegovy and Zepbound Debunked. Healthline, July 17, 2024. Accessed at www.healthline.com/health-news/wegovy-zepbound-myths-debunked.
- Grutzik, R. UVA Health Weighs In: 5 Misconceptions About Weight Loss Drugs. University of Virginia Today, Jan. 19, 2024. Accessed at news.virginia.edu/content/uva-health-weighs-5-misconceptions-about-weight-loss-drugs.
- Wang, W, Volkow, ND, Berger, NA, et al. Association of Semaglutide with Risk of Suicidal Ideation in a Real-World Cohort. Nature Medicine, 2024 Jan5;30(1):168–176. Accessed at pmc.ncbi.nlm.nih.gov/articles/PMC11034947.
- Mills, D. GLP-1 Drugs Benefit Brain and Heart Health, But May Cause Kidney, GI Issues. Healthline, Jan. 21, 2025. Accessed at www.healthline.com/health-news/glp-1-drugs-benefits-risks-healthoutcomes.
- Fischer, K. Struggling to Lose Weight on Obesity Drugs? Here’s What You Need to Know. Health, Nov. 8, 2024. Accessed at www.health.com/weight-loss-drugs-ineffective-tips-8737773.
- National Association of Insurance Commissioners. Does Insurance Cover Prescription Weight Loss Injectables? July 31, 2024. Accessed at content.naic.org/article/does-insurance-cover-prescription-weightloss-injectables.
