IVIG Plus Glucocorticoids Superior to IVIG Alone for MIS-C Associated with COVID-19: Review and Meta-Analysis

A new systematic review and meta-analysis conducted by U.S. and Nepalese collaborators supports the use of IVIG with glucocorticoids compared to IVIG alone.
Scientists Are Developing Cheaper, Patent-Free and Easier-to-Make COVID Vaccine

Researchers at the Texas Children’s Hospital Center for Vaccine Development at Baylor College of Medicine are developing a COVID-19 vaccine using a conventional method that will make the production and distribution cheaper and more accessible.
New Drugs Approved by FDA to Treat Eczema
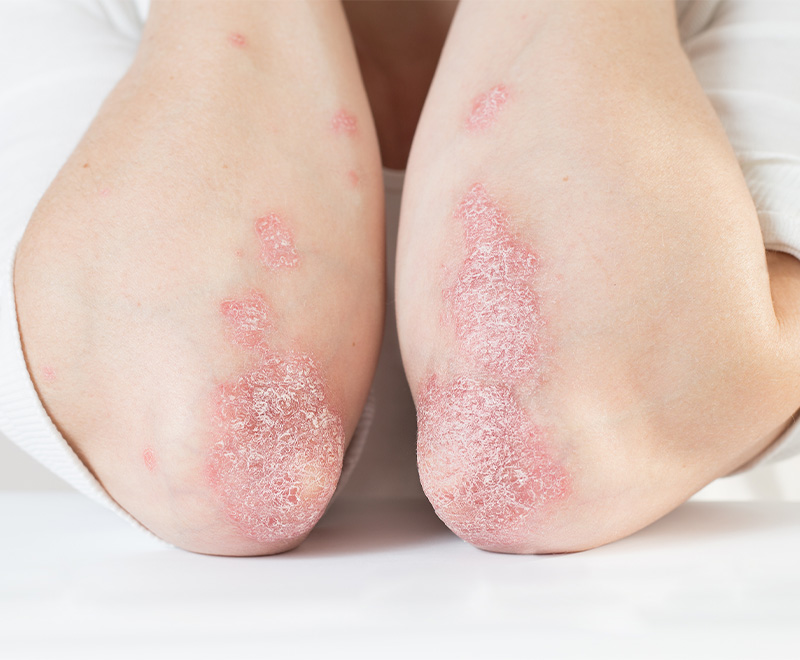
AbbVie’s Rinvoq and Pfizer’s Cibinqo have been approved by the U.S. Food and Drug Administration (FDA) to treat moderate-to-severe atopic dermatitis.
Virus Identified as Cause of Multiple Sclerosis
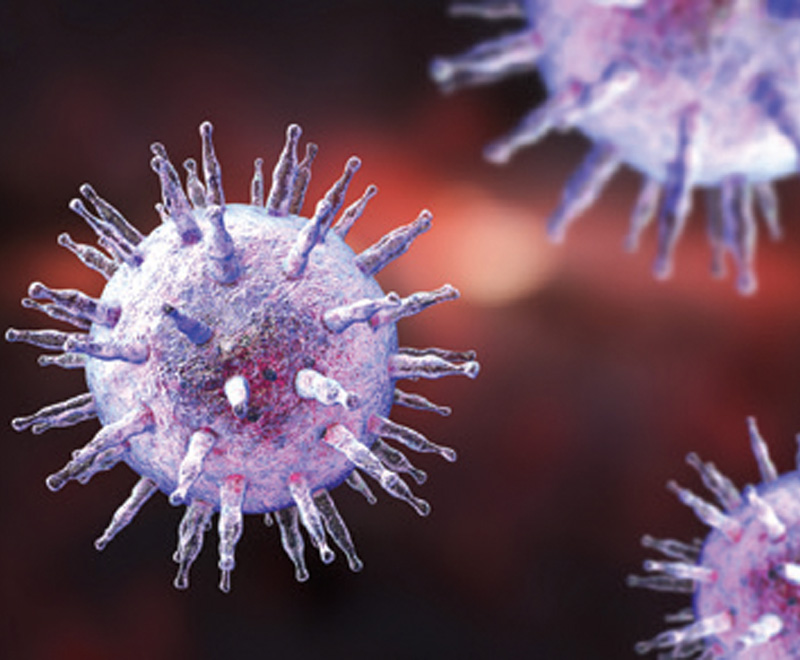
A new study shows the Epstein-Barr virus (EBV) is essential for the development of multiple sclerosis (MS).
FDA Approves Argenx’s Vyvgart to Treat MG
The U.S. Food and Drug Administration (FDA) has approved Vyvgart (efgartigi-mod) to treat generalized myasthenia gravis (gMG) in adults who test positive for the anti-acetylcholine receptor (AChR) antibody.
Two Major Changes in Medicare in 2022

The COVID-19 pandemic highlighted gaps the healthcare system. Two improvements to Medicare in 2022 seek to give people greater access to telehealth and other care delivery options.
Researchers at Emory University Awarded Grant to Pursue a Cure for HIV
An Emory University-led research collaboration has been awarded a five-year $23.8 million grant from the National Institutes of Health (NIH) to fast-track research to cure HIV infection or put it in permanent remission.
HHS Study Shows 63-Fold Increase in Medicare Telehealth Utilization During the Pandemic

A report from the U.S. Department of Health and Human Services (HHS) has found that considerable increases in the use of telehealth helped maintain some healthcare access during the COVID- 19 pandemic,
FDA Approves Novartis’ Cosentyx to Treat Arthritis in Children and Adolescents

Novartis has received U.S. Food and Drug Administration (FDA) approval for its Cosentyx (secukinumab) to treat children and adolescents with enthesitis-related arthritis and psoriatic arthritis.
Many Clinically Stable CIDP Patients Can Safely Stop IVIG Maintenance Treatment

While intravenous immune globulin (IVIG) therapy is efficacious for patients with chronic inflammatory demyelinating polyneuropathy (CIDP), the lack of biomarkers for disease activity makes the need for ongoing treatment difficult to assess.
