IVIG Improves Live Birth Rate in Women with Immune Conditions and Recurrent Pregnancy Loss: Review and Meta-Analysis

Dutch investigators conducted a systematic review and meta-analysis of studies on the effectiveness of IVIG treatment of this specific population.
IVIG Plus Glucocorticoids Superior to IVIG Alone for MIS-C Associated with COVID-19: Review and Meta-Analysis

A new systematic review and meta-analysis conducted by U.S. and Nepalese collaborators supports the use of IVIG with glucocorticoids compared to IVIG alone.
New Drugs Approved by FDA to Treat Eczema
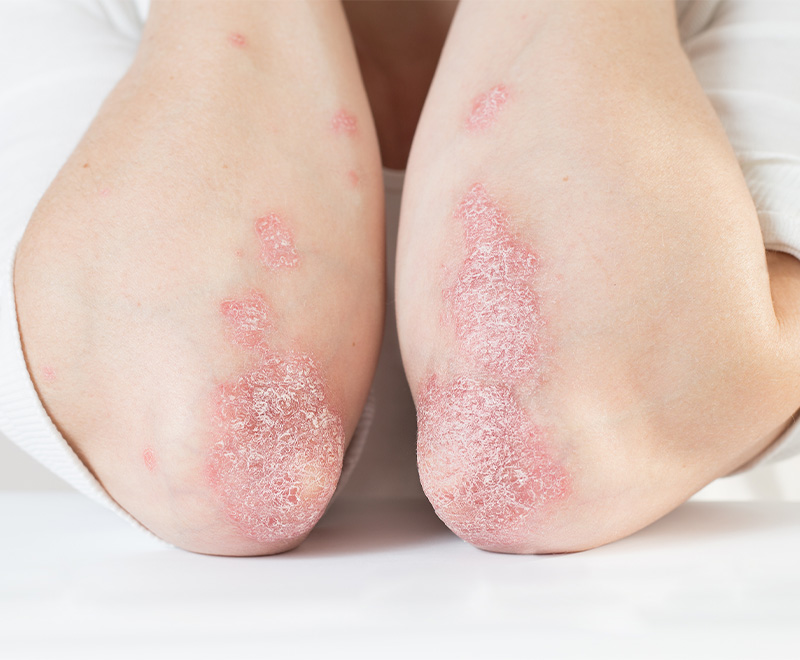
AbbVie’s Rinvoq and Pfizer’s Cibinqo have been approved by the U.S. Food and Drug Administration (FDA) to treat moderate-to-severe atopic dermatitis.
FDA Approves Novartis’ Cosentyx to Treat Arthritis in Children and Adolescents

Novartis has received U.S. Food and Drug Administration (FDA) approval for its Cosentyx (secukinumab) to treat children and adolescents with enthesitis-related arthritis and psoriatic arthritis.
Many Clinically Stable CIDP Patients Can Safely Stop IVIG Maintenance Treatment

While intravenous immune globulin (IVIG) therapy is efficacious for patients with chronic inflammatory demyelinating polyneuropathy (CIDP), the lack of biomarkers for disease activity makes the need for ongoing treatment difficult to assess.
Addition of Eltrombopag Improves Efficacy of Standard Therapy for Aplastic Anemia
Eltrombopay found to improve therapy for aplastic anemia.
IG Treatment May Reduce Acute Exacerbations of COPD
Observational studies suggest immune globulin (IG) treatment may reduce the frequency of acute exacerbations of chronic obstructive pulmonary disease (AECOPD).
IVIG Plus Glucocorticoids Effective for Treating COVID-19 Pediatric Syndrome

A large multicenter clinical trial has found intravenous immune globulin (IVIG) plus glucocorticoids may be better than IVIG alone for treating multisystem inflammatory syndrome in children (MIS-C) caused by COVID-19.
Kedrion to Market RYPLAZIM to Treat Rare Disease

Kedrion Biopharma is now marketing and distributing RYPLAZIM (plasminogen human-tvmh) in the United States to treat C-PLGD.
FDA Approves Avacopan to Treat Rare Autoimmune Disease

The U.S. Food and Drug Administration (FDA) has approved Chemo- Centryx Inc.’s Avacopan, sold under the brand name Tavneos, to treat antineutrophil cytoplasmic antibody-associated vasculitides.
